Precision Biologics Pipeline – Anticancer Drugs Targeting O-glycans
IMPORTANCE OF TARGETING O-GLYCANS SPECIFICALLY EXPRESSED BY CANCER CELLS:
- Glycosylation involves post-translational modifications of mammalian cells essential for their function. One glycosylation pattern often disrupted in cancer cells is O-glycosylation.
- During early stages of cancer a disruption of the normal synthesis of O-glycans, attached to proteins of normal cells, often occurs. This phenomenon leads to the expression of truncated O-glycans on the proteins of cancer cells.
- Truncated O-glycans are expressed specifically by several types of cancers, including solid tumors of epithelial origin and some hematological malignancies (acute myeloid leukemia and multiple myeloma). Aberrant expression of truncated O-glycans by cancer cells is associated with cancer growth, progression, invasion and metastasis.
- Truncated O-glycans are not expressed in healthy cells or tissues.
- The employment of anti-cancer therapy with strong binding affinity for cancer cells carrying truncated O-glycans, could represent a novel and promising tool in treating both solid and liquid tumors.
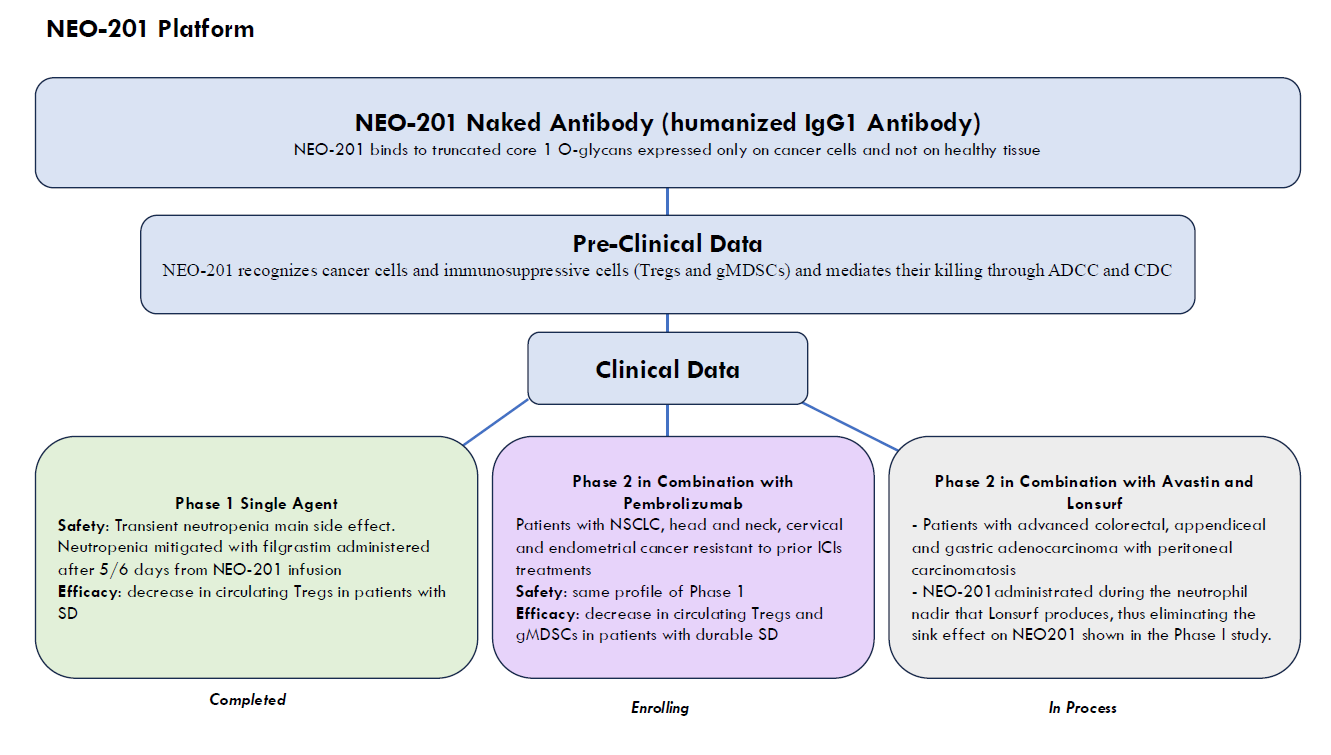
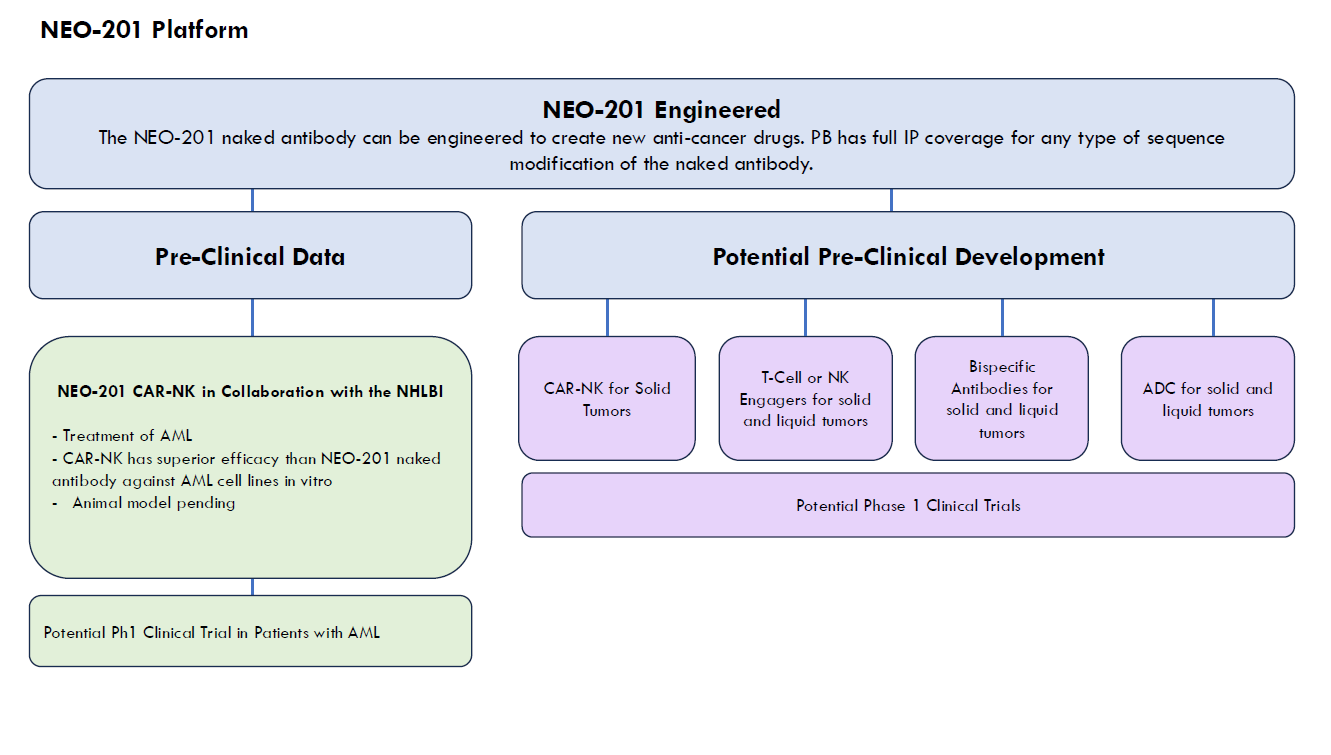
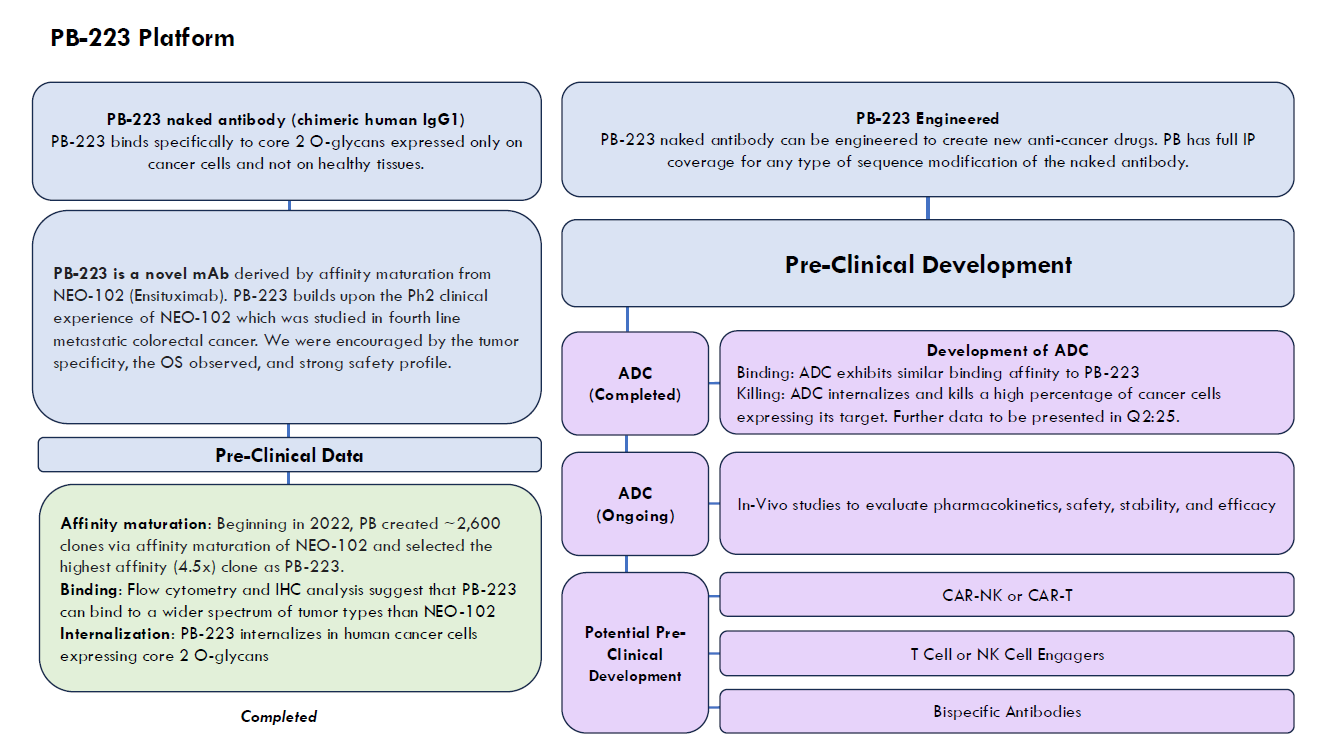
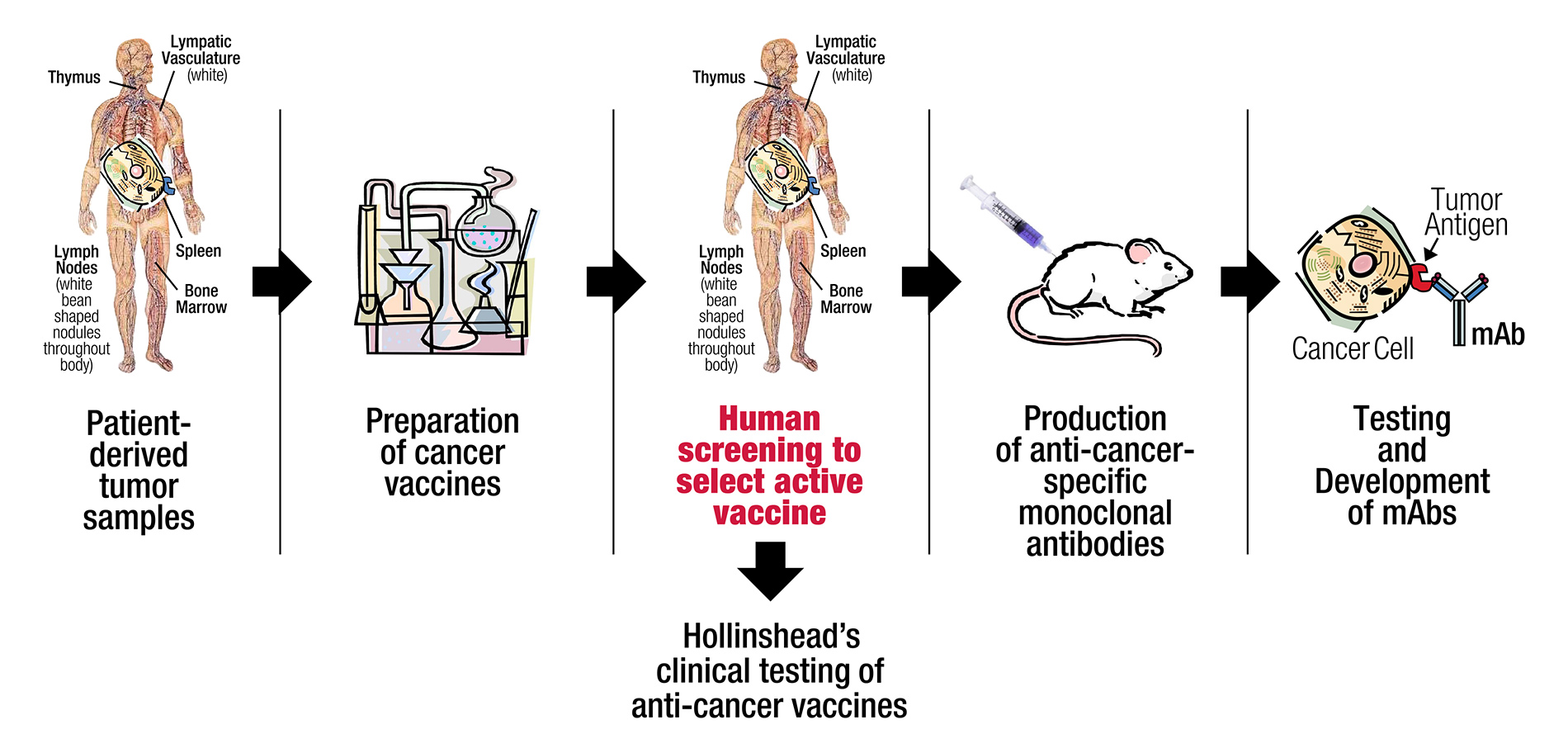
Utilizing a unique discovery platform of cancer vaccines, based on decades of basic research by a distinguished lead pioneer in immunotherapy, Precision Biologics has developed a pipeline of novel monoclonal antibodies (mAbs) for numerous types of cancer.